
California Mouse
Distribution, Abundance, and Seasonality
Found from San Francisco Bay south and east along the Coast Ranges to the border of Mexico, and at lower elevations in the western Sierra Nevada from Mariposa Co. south to Kern Co. Common resident of montane, mixed, and chamise-redshank chaparral, coastal scrub, valley foothill hardwood, and valley foothill hardwood-conifer habitats. Also found in a variety of other wooded habitats. Generally found below 1600 m (5000 ft), but may be found up to 2800 m (8000 ft).
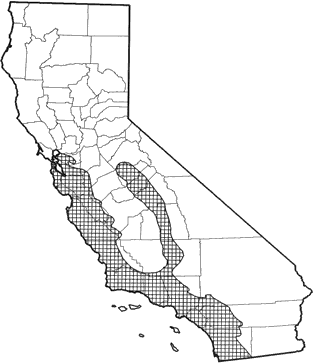
Range Map
Specific Habitat Requirements
Feeding: Major foods include fruits, flowers and seeds of a variety of plants, fungi, and arthropods. In woodland habitats, acorns are eaten, but the seeds of California bay are the major food (Merritt 1974). Laboratory studies have shown that P. californicus is capable of cracking the seed coats of California bay, while P. truei cannot (Merritt 1974). In coastal scrub, the California mouse prefers the seeds and fruits of shrubs, especially Salvia (Meserve 1976b). A good climber, and forages extensively in shrubs as well as on the ground (Meserve 1977). Food sometimes is cached.
Cover: Occupied or abandoned woodrat nests are an important source of cover, and there is experimental evidence that California mouse density is related to the availability of woodrat nests (Cranford 1982). Other suitable forms of cover are rotten logs, litter, stumps, and snags, any of which may be the site of a large nest with a central chamber. This species seems to be a relatively inefficient burrower.
Reproduction: An elaborate nest of grasses and forbs is built in a burrow, woodrat den, dense brush, or debris.
Water: Probably obtains water from vegetation and dew. In lab, California mice drank 0.11 ml/g/day (Merritt 1974). The California mouse is not an efficient water conserver compared to other rodents in its habitat (MacMillen 1964).
Pattern: Prefers dense oak-bay woodland or mature chaparral or coastal scrub interspersed with grassy areas. Density is related to presence of woodrat nests and, in woodlands, density of California bay. May invade dry habitats seasonally.
Species Life History
Activity Patterns: Yearlong nocturnal activity. There may be an activity peak just before dawn. May enter torpor in periods of food shortage on a daily basis (Hudson 1967).
Seasonal Movements / Migration: May invade dry habitats in spring and early summer.
Home Range: MacMillen (1964) reported home ranges of 0.15 ha (0.37 ac). The home range size of this species may be underestimated if arboreal activity is not considered (Meserve 1977). Merritt (1974) reported densities of 77 and 92/ha (35 and 42/ac) in xeric chaparral-oak woodland and mesic oak-laurel woodland, respectively. Typical densities ranged from 9-27/ha (4-11/ac) (Merritt 1978). Smith (1981) reported that numbers of P. californicus remained at fairly constant low levels over a 9 yr period.
Territory: Mainly non-territorial, but like other Peromyscus shows aggressive behavior near the nest (McCabe and Blanchard 1950).
Reproduction: Mating may be yearlong, but occurs mostly from March to September. Gestation is about 21-25 days (Svihla 1932). Most young are born from April through October. Litter size averages 2 (range 1-3). There may be 3-4 litters per yr (McCabe and Blanchard 1950) in the wild, and up to 6 litters per yr in lab. The young are somewhat precocious compared to other Peromyscus, but have a relatively long period to weaning (5 wk), and require a long period to reach sexual maturity (at least 11 wk for females, longer for males) (McCabe and Blanchard 1950). There is a slight increase in litter size with age of the female. The male may assist in care of the young.
Niche: In foraging behavior, this may be the most specialized of the California Peromyscus species. California mice apparently competitively exclude P. truei from woodrat nests. The California mouse is largely sympatric with Neotoma fuscipes and P. truei. Fire favors P. maniculatus over P. californicus. Predators include weasels, coyotes, owls, snakes, and feral cats.
Sources & References
California Department of Fish and Game, 1999.
California's Wildlife, Sacramento, CA.
Written by: P. Brylski, J. Harris, reviewed by: H. Shellhammer, edited by: R. Duke, S. Granholm
Cranford, J. A. 1982. The effect of woodrat houses on population density of Peromyscus. J. Mammal. 63:663-666. Hudson, J. W. 1967. Variations inthe patterns of torpidity of small homoiotherms. Pages 30- 46 in K. C. Fisher, C. P. Lyman, E. Schonbaum, and F. E. South, Jr. eds. Proc. Third Int. Symp. Mammal. Hibernation. Elsevier, New York. 535pp. Layne, J. N. 1968. Ontogeny. Pages 148-253 in J. A. King, ed. Biology of Peromyscus. Am. Soc. Mammal. Special Publ. No. 2. 593pp. MacMillen, R. E. 1964. Population ecology, water relations and social behavior of a southern California semidesert rodent fauna. Univ. Calif. Publ. Zool. 71:1-59. McCabe, T. T., and B. D. Blanchard. 1950. Three species of Peromyscus. Rood Assoc., Santa Barbara, Ca. 136pp. Merritt, J. F. 1974. Factors influencing the local distribution of Peromyscus californicus in northern California. J. Mammal. 55:102-114. Merritt, J. F. 1978. Peromyscus californicus. Mammal Species No. 85. 6pp. Meserve, P. L. 1976a. Habitat and resource utilization by rodents of a California coastal sage scrub community. J. Anim. Ecol. 45:647-666. Meserve, P. L. 1976b. Food relationships of a rodent fauna in a California coastal sage scrub community. J. Mammal. 57:300-319. Meserve, P. L. 1977. Three-dimensional home ranges of cricetid rodents. J. Mammal. 58:549-558. Smith, M. F. 1981. Relationships between genetic variability and niche dimensions among coexisting species of Peromyscus. J. Mammal. 62:273-285. Svihla, A. 1932. A comparative life history study of the mice of the genus Peromyscus. Univ. Mich. Mus. Zool., Misc. Publ. No. 24. 39pp.
California Animal Facts | California's Wildlife