Chisel-toothed Kangaroo Rat
Distribution, Abundance, and Seasonality
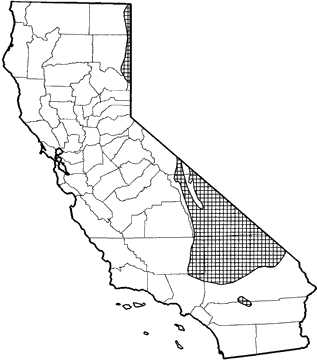
Occurs in the Owens Valley from Benton, Mono Co., south to Olancha, Inyo Co. Outside the Owens Valley, D. microps occurs from Death Valley southeast to Clark Mt., San Bernardino Co., as well as near Victorville in the Antelope Valley. It is found in northeastern California from Cedarville, Modoc Co., south to Beckworth Pass, Plumas Co. Throughout much of its range, it is found primarily in sagebrush and desert scrub habitats, and to a lesser extent in pinyon-juniper habitat.
Range Map
Specific Habitat Requirements
Feeding: Feeds on leaves of shrubs and forbs and on seeds of grasses and forbs. It specializes on the leaves of Atriplex confertifolia. Fresh leaves are harvested by climbing into shrubs (Kenagy 1972). Entire Atriplex leaves are consumed during the spring, but during the rest of the year only the inner, less saline tissues are utilized. The lower incisors are chisel-shaped and seem to be adapted for shearing off the salty outer layers of the leaves (Kenagy 1972). Seeds are gathered in the open between shrubs; fresh leaves are harvested from the shrub canopy.
Cover: Beatley (1976a) reported that disturbance at the Nevada Test Site, which reduced canopy cover of shrubs, resulted in recolonization by D. merriami, but not by D. microps. Burrows are constructed in sandy or gravelly soils. Steep slopes are avoided. The distribution and abundance of D. microps seem to be limited by the abundance of preferred shrubs with succulent and palatable leaves (Kenagy 1973a).
Reproduction: Burrows typically established in hillocks under shrubs, e.g., greasewood in Owens Valley (Kenagy 1973a). Depth and complexity of burrow systems vary considerably with the type of soil. Nest usually is in deepest part of burrow, with both nests and food caches of Atriplex leaves found within the same burrows (Anderson and Allred 1964).
Water: Cannot survive on dry seeds; must have some source of water (Kenagy 1972, 1973b). Its association with areas of higher rainfall in Nevada (Beatley 1976b) and its reliance upon green vegetation in California (Kenagy 1972) suggest D. microps has a high body-water turnover rate. It ranks lowest among North American Heteromyidae in ability to concentrate urine.
Pattern: Moderately dense desert scrub with sandy or gravelly substrate. Beatley (1976a) described a strong correlation between the occurrence of D. microps and high shrub cover. Soil characteristics varied widely, from upland habitats of sand mixed with boulders, cobbles, and gravel, to lowland alluvial habitats of silty and sandy loams (Beatley 1976a).
Species Life History
Activity Patterns: Active year-round nocturnally. Above-ground activity begins early in night and decreases through night (Kenagy 1973a, O'Farrell 1974, Kenagy 1976). Onset of activity synchronized, but variation in time of cessation is considerably greater. In winter, activity pattern is trimodal, but during rest of year activity remains high and stable throughout night (O'Farrell 1974). Inclement weather inhibits activity (Kenagy 1973a, O'Farrell 1974). Moonlight has no effect on onset or termination of activity periods (Kenagy 1973a), but it may inhibit amount of activity (O'Farrell 1974).
Seasonal Movements / Migration: None.
Home Range: Home ranges of 0.33-1.23 ha (0.8-3.0 ac) were reported in southern Nevada,1.1-3.1 ha (2.75-7.75 ac) for male D. microps and 0.4-5.2 ha (1-13 ac) for females in the Mojave Desert (Maza et al. 1973). Allred and Beck (1963b), working in Nevada, found movements to average 70-75 m (225-250 ft).
Territory: No data found, but probably equal to home range.
Reproduction: In California, breeds from January to June. Gestation is 33 days (Eisenberg and Isaac 1963). Litter size averaged 2.3 (range 1-4) in Nevada (Hall 1946). Young are weaned at 24-33 days (Chew and Butterworth 1959).
Niche: Known, or suspected, predators include: kit foxes, red racers, long-nosed snakes, and leopard lizards (French et al. 1967).
Sources & References
California Department of Fish and Game, 1999.
California's Wildlife, Sacramento, CA.
Written by: P. Brylski, reviewed by: H. Shellhammer, edited by: R. Duke
Allred, D. M., and D. E. Beck. 1963a. Ecological distribution of some rodents at the Nevada atomic test site. Ecology. 44:211-214. Allred, D. M., and D. E. Beck. 1963b. Range of movement and dispersal of some rodents at the Nevada atomic test site. J. Mammal. 44:190-200. Anderson, A. O., and D. M. Allred. 1964. Kangaroo rat burrows at the Nevada test site. Great Basin Nat. 24:93-101. Beatley, J. C. 1976a. Environments of kangaroo rats (Dipodomys) and effects of environmental change on populations in southern Nevada. J. Mammal. 57:67-93. Beatley, J. C. 1976b. Rainfall and fluctuating plant populations in relation to distributions and numbers of desert rodents in southern Nevada. Oecologia (Berlin) 24:21-42. Chew, R. M., and B. B. Butterworth. 1959. Growth and development of Merriam's kangaroo rat, Dipodomys merriami. Growth 23:75-95. Eisenberg, J. F., and D. E. Isaac. 1963. The reproduction of heteromyid rodents in captivity. J. Mammal. 44:61-67. French, N. R., B. G. Maza, and A. P. Aschwanden. 1967. Life spans of Dipodomys and Perognathus in the Mojave Desert. J. Mammal. 48:537-548. Hall, E. R. 1946. Mammals of Nevada. Univ. California Press, Berkeley. 710pp. Kenagy, G. J. 1972. Saltbush leaves: excision of hypersaline tissue by a kangaroo rat. Science 178:1094-1096. Kenagy, G. J. 1973a. Daily and seasonal patterns of activity and energetics in a heteromyid rodent community. Ecology 54:1201-1219. Kenagy, G. J. 1973b. Adaptations for leaf eating in the Great Basin kangaroo rat, Dipodomys microps. Oecologia (Berlin) 12:383-412.. Kenagy, G. J. 1976. The periodicity of daily activity and its seasonal changes in free-ranging and captive kangaroo rats. Oecologia (Berlin) 24:105-140. Maza, B. G., N. R. French, and A. P. Aschwanden. 1973. Home range dynamics in a population of heteromyid rodents. J. Mammal. 54:405-425. O'Farrell, M. J. 1974. Seasonal activity patterns of rodents in a sagebrush community. J. Mammal. 55:809-823.
California Animal Facts | California's Wildlife